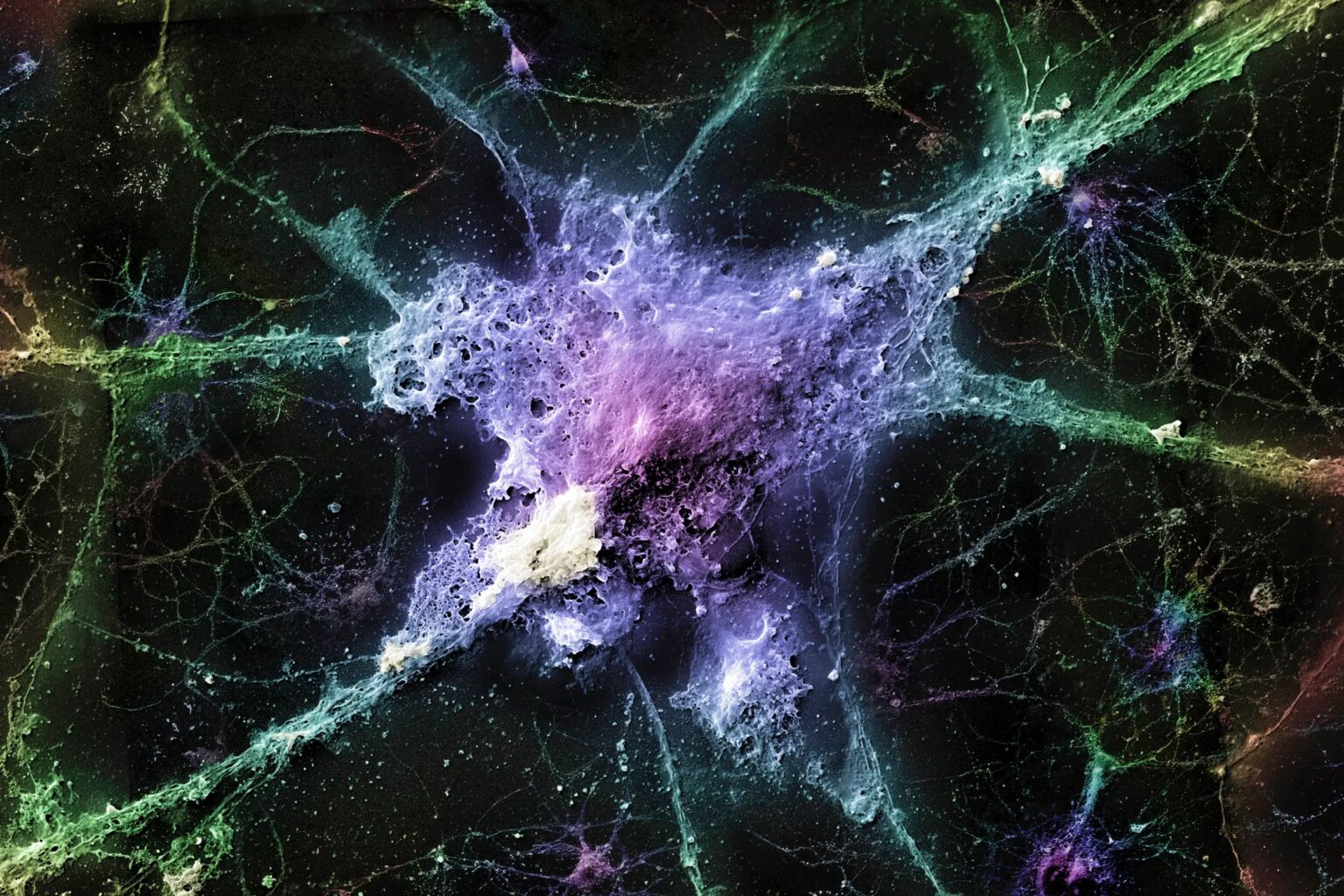
Scanning electron micrograph of mouse nerve cells affected by misfolded amyloid and beta proteins, believed to cause Alzheimer’s disease.
Linnea Lundgren/Linear Imaging/Science Photo Library
Research is increasingly focused on utilizing the brain’s waste disposal system to potentially slow or mitigate Alzheimer’s disease. A recent technique has demonstrated success in removing toxic protein aggregates associated with Alzheimer’s from mouse brains, leading to improved memory and learning test results.
This technique targets a receptor known as DDR2, traditionally associated with lung health. “Inhibiting the DDR2 pathway could theoretically decrease amyloid beta protein levels while simultaneously enhancing waste removal,” explains Jia Li from Guangzhou Medical University, China. “We are optimistic that we can ultimately reverse Alzheimer’s disease.”
The buildup of misfolded proteins, such as amyloid plaques and tau tangles in the brain, is considered a primary trigger for Alzheimer’s. While existing medications can remove amyloid aggregates, they often do not significantly alleviate symptoms. Thus, research is shifting towards innovative strategies, including enhancing the glymphatic system responsible for waste clearance in the brain.
Lee and colleagues plan to further investigate receptors in cell membranes that may boost glymphatic function as one of their roles. DDR2, studied extensively for its role in pulmonary fibrosis, is also implicated in Alzheimer’s disease by Jinsu and his team at Guangzhou Medical University. Pulmonary fibrosis occurs when the extracellular matrix surrounding cells fails, leading to excessive collagen deposition and oxygen supply limitations.
Research indicates that the malfunctioning extracellular matrix is associated with amyloid and tau proteins in Alzheimer’s disease. “This restriction of oxygen can hinder cognitive functions like thinking and memory,” states Lee.
To explore DDR2’s role, the researchers reviewed human tissue databases and discovered DDR2’s scarcity. However, they found substantial amounts in brain samples from Alzheimer’s patients. “We confirmed that DDR2 is prevalent in Alzheimer’s disease brain tissue for the first time,” notes Su.
Through various experiments in human and primate cells, along with mouse models, researchers propose that DDR2 regulates the cellular dysfunction responsible for the disease’s symptoms. This is substantiated by findings that three cell types increase DDR2 in their membranes during Alzheimer’s: reactive astrocytes, surrounding amyloid beta masses; perivascular fibroblasts, which alter activity prior to Alzheimer’s onset; and choroid plexus epithelial cells that are crucial for cerebrospinal fluid production, essential for the glymphatic system.
These findings suggest that targeting DDR2 could impact multiple facets of Alzheimer’s simultaneously, as noted by Siju Gu from Harvard University. Yet, due to the complexity of the condition, he remains cautious about potential reversibility of Alzheimer’s disease.
The researchers developed a monoclonal antibody aimed at blocking the DDR2 receptor. In mouse models of Alzheimer’s, this intervention improved spatial learning and memory, alongside reduced DDR2 levels, fewer amyloid plaques, and enhanced glymphatic activity.
“The mouse model results are promising and highlight the role of glymphatic function and cerebrospinal fluid dynamics in brain health,” Gu remarked. “This suggests DDR2 could be a viable target for Alzheimer’s disease therapies.”
Cesar Cunha from Denmark’s Novo Nordisk Foundation Center for Basic Metabolic Research expressed appreciation for the researchers’ focus on more than just amyloid plaques, noting their model relates to a rare inherited form of Alzheimer’s that typically arises earlier. Its applicability to the more common late-onset Alzheimer’s remains uncertain.
Professor Hsu, however, indicates that DDR2 upregulation occurs in both familial and late-onset Alzheimer’s, suggesting the treatment has potential widespread efficacy. DDR2 expression appears to increase with age, a factor alongside hypoxia, both recognized risk factors for late-onset Alzheimer’s.
Currently, researchers are embarking on clinical trials that use tracers to monitor DDR2 levels in Alzheimer’s patients’ brains, aiming to determine the antibodies’ delivery paths. They are also developing smaller antibodies to facilitate more efficient crossing of the blood-brain barrier.
Topic:
Source: www.newscientist.com