Like many children affected by Hunter syndrome, Cole is missing a crucial enzyme required to break down specific molecules. As these toxins accumulate, they wreak havoc on vital organs, particularly the heart and brain, often resulting in dementia-like symptoms. This genetic disorder, also referred to as mucopolysaccharidosis type II (MPS II), impacts around 500 individuals in the United States, predominantly boys.
Experts believe the recently approved drug by Denali Therapeutics, an intravenous enzyme replacement therapy, could revolutionize treatment options, especially since current standard care only addresses the physical manifestations of the disease. Denali’s innovative therapy also aims to combat cognitive decline.
While new medications can’t reverse existing regression, they hold potential to extend a child’s life and stave off numerous symptoms when administered early.
Alison M. Donnelly of NBC News
“If we can treat very young children before irreparable damage occurs, the possibilities are nearly limitless,” says Dr. Joseph Muenzer of the Muenzer MPS Research and Treatment Center at the University of North Carolina at Chapel Hill, who specializes in Hunter syndrome and other rare mucopolysaccharide disorders.
“I can’t predict how they will fare in the future, but it will certainly be different,” he remarked.
Before regression began, Cole learned to read and spoke in complete sentences. Gradually, his speech deteriorated, leaving him only able to say “Mommy” before losing all ability to communicate. Despite being a teenager now, he is developmentally similar to a 3-year-old.
The FDA’s approval of Denali’s drug was a significant relief for families with children suffering from Hunter syndrome and for the broader rare disease community. Recently, the FDA faced criticism for denying several promising treatments for rare conditions, prompting advocacy groups to stage protests and advocate for more supportive policies.
In an email to NBC News, the FDA noted that the number of drug approvals and denials under its current administration aligns with historical trends from the past decade. FDA Commissioner Dr. Marty McCurry hailed Denali’s approval as “a milestone day for children combating Hunter syndrome and their families,” pledging continued efforts to expedite treatments for rare diseases.
Many healthcare professionals, including Munzer, who led the Denali trial, are hopeful that advancements in treatments will continue.
“These are devastating diseases,” Munzer emphasized. “Their rarity should not lead to neglect.”
Denali’s drug, known as Avraya, marks the first FDA-approved treatment for Hunter syndrome in the United States in two decades and is the first to penetrate the blood-brain barrier, targeting the neurological complications associated with the disease.
Experts like Müntzer assert that Avraya could significantly extend life expectancy, based on encouraging human trial data. A clinical trial illustrated significant reductions in important disease biomarkers in the cerebrospinal fluid after just 24 weeks, with 93% of pediatric participants achieving levels akin to those without Hunter syndrome.
For countless families dealing with this condition, the approval of this drug signifies more than a new treatment; it acknowledges the inherent value of every child.
Laurent Jaskalski, a 6-year-old from New Berlin, Wisconsin, was diagnosed with Hunter syndrome at age four. He’s always struggled with communication, but his mother, Kylie Jaskalski, fears that his speech difficulties may lead others to underestimate his capabilities. This is a painful misconception about a joyful child who loves to cuddle and eagerly anticipates going to school each day.
“He brings immense joy,” Jaskalski said. “He spreads happiness and peace to everyone he meets.”
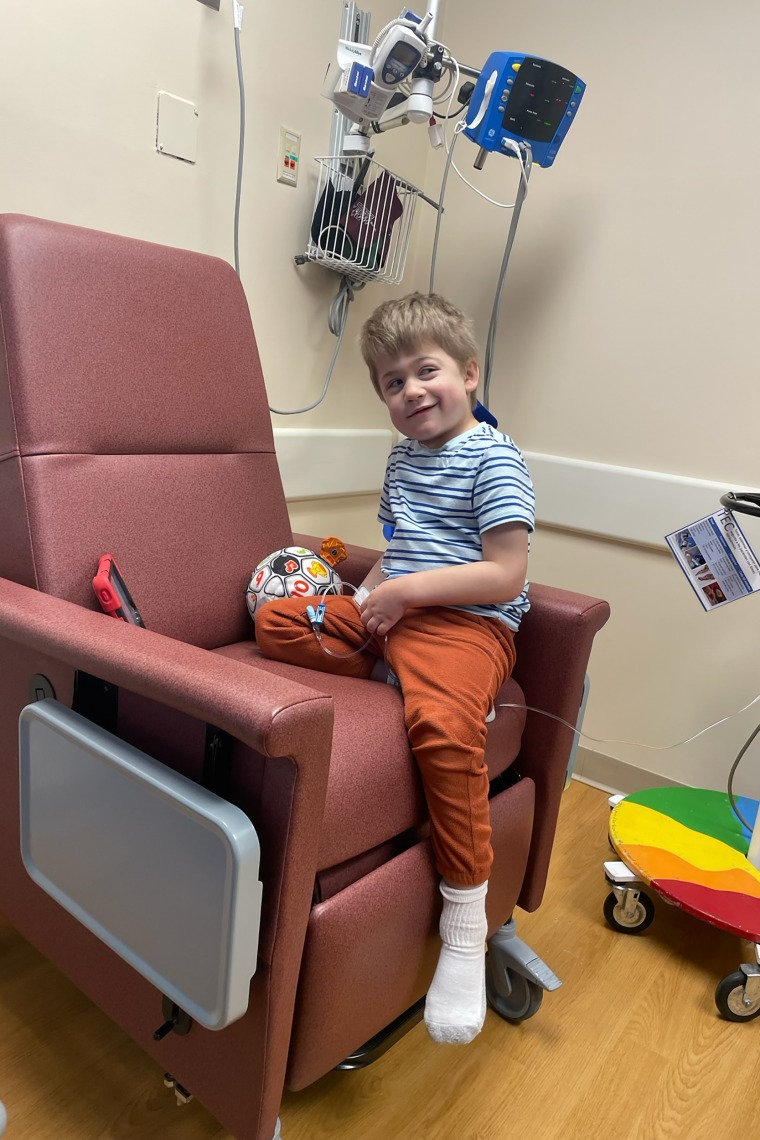
Provided by Kylie Jaskalski
Initially, Jaskalski felt devastated when Laurent was diagnosed, but she later found it even more heart-wrenching to witness her son’s deterioration.
If insurance permits access to Denali’s drug for Laurent, “we might not have to remain passive,” she expressed.
Currently, Laurent receives weekly infusions of Elaplus (FDA-approved in 2006), which is the prevailing standard treatment. While this IV therapy isn’t flawless, it has stabilized his physical decline. Recently, Jaskalski has observed weakness and discomfort on Laurent’s left side, making walking challenging. He also suffers from mild hearing loss.
Recent advancements in newborn screening for Hunter syndrome across states like California to Rhode Island indicates a promising future, as early detection allows for timely intervention with new drugs, potentially mitigating cognitive decline.
Children with Hunter syndrome often face severe health challenges and a life expectancy ranging from 10 to 20 years. Those with the non-neurological variant may reach adulthood but still endure progressive physical complications, primarily affecting their airways and heart.

Provided by Christina Caldwell
The introduction of new treatments is especially hopeful for families with children across the spectrum of Hunter syndrome. Christina Caldwell’s 3-year-old grandson, Kashton Estes, who has the non-cognitive variant, undergoes IV infusions of Elaplus, which he affectionately calls “Spider-Man juice.” Thanks to this treatment, he enjoys running, jumping, and riding his bike.
Caldwell is eager for Kashton to be treated with Denali’s new drug as two of his cousins also have Hunter syndrome, and the family wishes to prevent future health complications for all three children.
“We’re not asking for much. Just provide us with the medication to keep our children alive,” she stated.
Regarding insurance coverage, Denali Therapeutics stated it has engaged in “constructive” dialogues with payers and is prioritizing swift access to Avraya for families dealing with Hunter syndrome. Currently, the drug is approved only for pediatric patients, but the company is working on expanding its clinical data to include young adults. Denali also aims to leverage its blood-brain barrier technology for various other diseases, including neurodegenerative disorders.
Ms. Stevens, the mother of 15-year-old Cole, eagerly anticipates administering the new medication to her son. She has devoted her life to aiding not just Cole but others in similar situations. In 2022, she took on the role of executive director at Munzer’s MPS Research and Treatment Center at UNC-Chapel Hill.
Upon learning last week about the FDA’s early approval for the new drug, Stevens rushed through the treatment center, sharing the news with patients and staff. There were tears of joy and hugs all around.
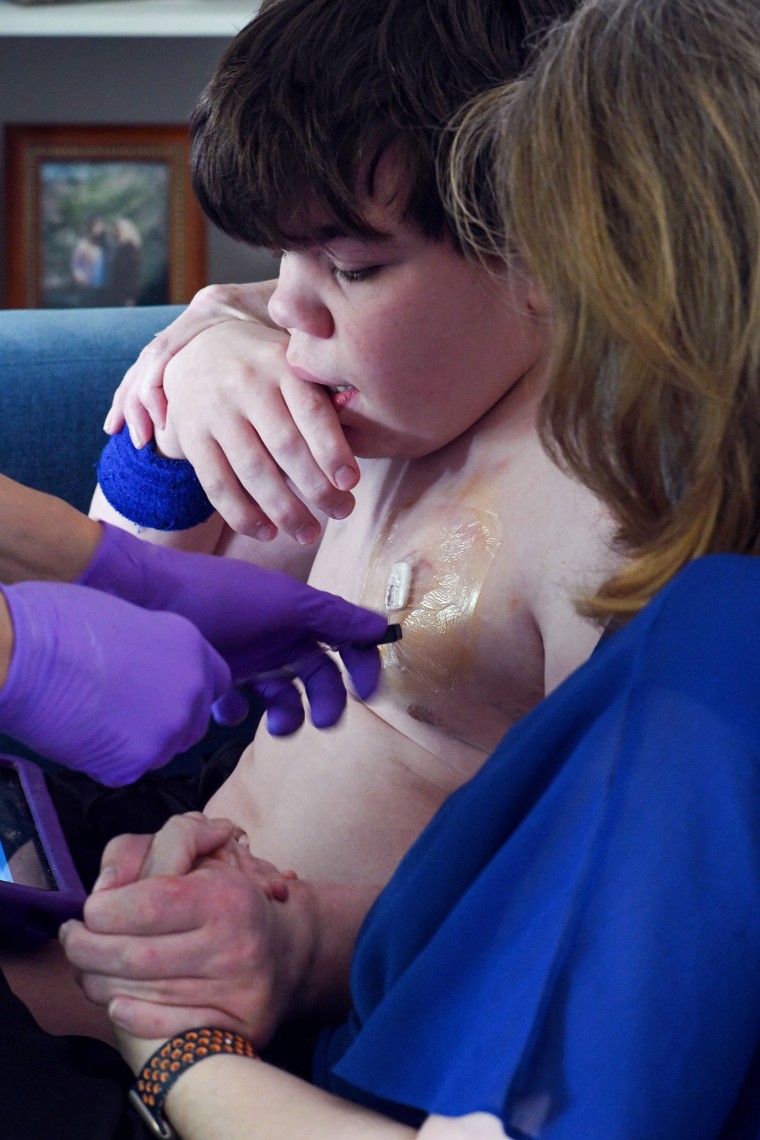
Alison M. Donnelly of NBC News
Stevens understands that Denali’s treatment cannot reverse Cole’s previous regression, meaning he may not attend university or secure a job. However, she remains profoundly grateful.
“My hope is for him to maintain stability,” Stevens expressed. “Even with a progressive disease, achieving stability is a victory.”
Source: www.nbcnews.com