The history of cancer can be traced back to ancient Egyptian civilizations, where it was thought to be a divine affliction. Over the years, great strides have been made in understanding cancer’s causes and exploring diverse treatment options, although none have proven to be foolproof. Recently, a research team at Columbia University has pioneered a novel method for combating cancerous tumors by utilizing a combination of bacteria and viruses.
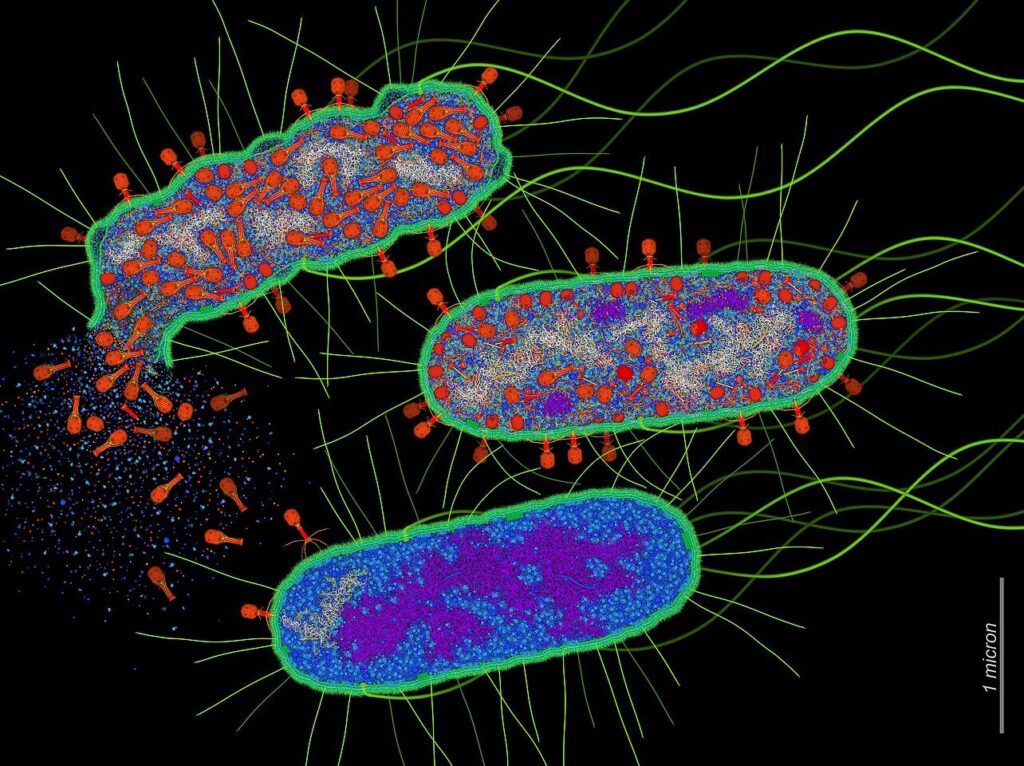
The researchers engineered this innovative strategy by infecting bacterial cells with Typhimurium that were modified to carry the Seneca virus A. The theory posited that when tumor cells engulf these bacteria, they would also take in the virus, which would then replicate within the cells, leading to their death and the subsequent distribution of the virus to surrounding cells. This technique has been termed Coordinated Activities of Prokaryotes and Picornaviruses for Safe Intracellular Delivery (CAPPSID).
Initially, the research team verified that Typhimurium was a suitable host for Seneca virus A. They infected a limited number of these bacteria with a modified variant of the virus that emitted fluorescent RNA. Subsequently, they applied a solution that facilitated viral entry into the bacteria. Using fluorescence microscopy, they confirmed the presence of viral RNA inside the bacterial cells, validating the infection. To further assist the viral RNA in escaping the bacteria and reaching cancer cells, the researchers added two proteins, ensuring that viral spread was contained to prevent infection of healthy cells.
After optimizing the bacteria and virus, the team tested the viral delivery system on cervical cancer samples. They found that viral RNA could replicate both outside of bacterial cells and inside cancer cells. Notably, newly synthesized RNA strands were identified within tumor cells, confirming the successful delivery and replication of the virus through the CAPPSID method.
Next, the researchers examined CAPPSID’s impact on a type of lung cancer known as small cell lung cancer (SCLC). By tracking fluorescent viral RNA within SCLC cells, they assessed the rate of viral dissemination post-infection. Remarkably, the virus continued to propagate at a consistent rate for up to 24 hours following the initial infection, demonstrating effective spread through cancerous tissue without losing vigor.
In a follow-up experiment, the researchers evaluated the CAPPSID method on two groups of five mice, implanting SCLC tumors on both sides of their backs. They engineered the Seneca virus A to generate a bioluminescent enzyme for tracking purposes and injected the CAPPSID bacteria into the tumors on the right side. Two days post-injection, the right-side tumor glowed, indicating active viral presence. After four days, the left-side tumor also illuminated, suggesting that the virus had successfully navigated throughout the mice’s bodies while sparing healthy tissues.
The treatment continued for 40 days, leading to complete tumor regression within just two weeks. Remarkably, upon observation over a subsequent 40-day period, the mice demonstrated a 100% survival rate, with no recurrence of cancer or significant side effects. The research team observed that the CAPPSID virus, being encapsulated by bacteria, could circumvent the immune response, thus preventing cancer cells from building immunity against it.
Finally, to prevent uncontrolled replication of Seneca virus A, the researchers isolated a gene from a tobacco virus responsible for producing an enzyme that activates a crucial protein in Seneca virus A. By incorporating this gene into the Typhimurium bacteria, they were able to independently produce this enzyme, ensuring the virus could not replicate or spread without the bacteria’s presence. Follow-up tests confirmed that this modified CAPPSID method improved viral spread while maintaining confinement within cancer-affected areas.
The research findings hold promising potential for the development of advanced cancer therapies. The remarkable regression of tumors in mice and the targeted delivery system of CAPPSID—without adverse effects—could lead to safer cancer treatments for human patients, eliminating the need for radiation or harmful chemicals. However, the researchers also cautioned about the risk of viral and bacterial mutations that may limit the effectiveness of CAPPSID and cause unforeseen side effects. They suggested that enhancing the system with additional tobacco virus-derived enzymes could help mitigate these challenges, paving the way for future research into innovative cancer therapies.
Post views: 148
Source: sciworthy.com