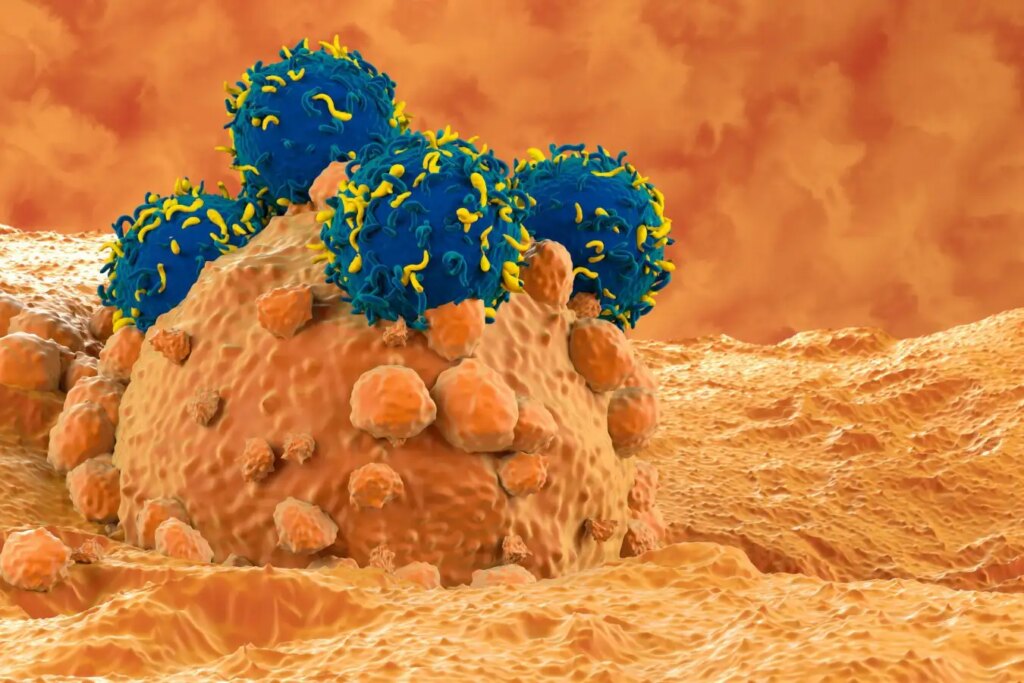
Illustation of CAR T cell therapy for melanoma, a form of skin cancer
Nemeth Laszlo/Shutterstock
Innovative therapies are transforming the treatment landscape for blood and skin cancers, with recent studies highlighting enhanced effectiveness. In murine models with advanced skin cancer, researchers have discovered that manipulating the physical properties of cancer cells amplifies the efficacy of immunotherapy—specifically, CAR T-cell therapy. This promising breakthrough could significantly improve survival rates for patients undergoing immunotherapy.
“This groundbreaking concept addresses a critical medical issue from a physical perspective,” notes Lee Sui from Queen Mary University of London, who is not associated with this research. “The outlook is very hopeful.”
Cancer cells are often softer when compared to healthy cells, which poses challenges. T cells, vital components of the immune system responsible for targeting cancer, sense environmental stiffness.
“We examined whether the softness of cancer cells allows them to evade the immune response and how T cell mechanosensing affects the cellular response to cancer,” explains Lee Tan, who presented his findings on May 11 at an academic conference hosted by the Swiss Federal Institute of Technology in Lausanne, Switzerland. The Biophysical Immunoengineering: From Insights to Clinical Applications conference in London focused on these innovative approaches.
The researchers set out to uncover why cancer cells exhibit softness by contrasting their membranes with those of healthy cells. They discovered that both murine and human cancer cells tend to be softer due to high cholesterol content in their membranes.
The team subsequently injected 24 mice with melanoma cells, notorious for being the deadliest skin cancer. Nine days post-injection, the mice received genetically modified T cells specifically designed to target the tumor, emulating CAR T-cell therapy, which is approved for conditions like acute lymphoblastic leukemia and B-cell lymphoma.
Additionally, the mice underwent three injections over five days of IL-15, a protein that heightens the cancer-killing capacity of tumor-specific T cells.
Crucially, only half of the mice received a third treatment involving methyl beta-cyclodextrin (meβCD), a compound that reduces cholesterol levels in cell membranes, administered directly into the tumors daily from day 9 to day 18 post-cancer cell injection. The other mice received saline as a control.
After roughly one month, all 12 mice that did not receive meβCD succumbed to rapidly-growing tumors. In stark contrast, only seven mice in the meβCD group perished, while five experienced complete tumor resolution. “The results are compelling. Very encouraging,” states Lance Cam from Columbia University, New York.
Further analysis indicated that meβCD enhanced the adherence of tumor-specific T cells to tumor cells by stiffening them. Consequently, T cells were more effective in delivering toxic agents such as perforin, which perforates and obliterates cancer cells.
The research team aims to extend this approach to a broader array of tumors in mice, according to Tang. “The significant challenge lies in ensuring this understanding translates to human applications,” Kam emphasizes. Few successful immune-targeting drugs in mice yield equivalent results in humans, primarily due to immune system disparities. However, since cancer cells tend to be soft in both species, there is potential for therapies that modify cancer cell stiffness to be more effective.
Moreover, researchers are actively working on developing therapeutics with effects akin to meβCD that can be delivered with a single injection.
Topics:
Source: www.newscientist.com