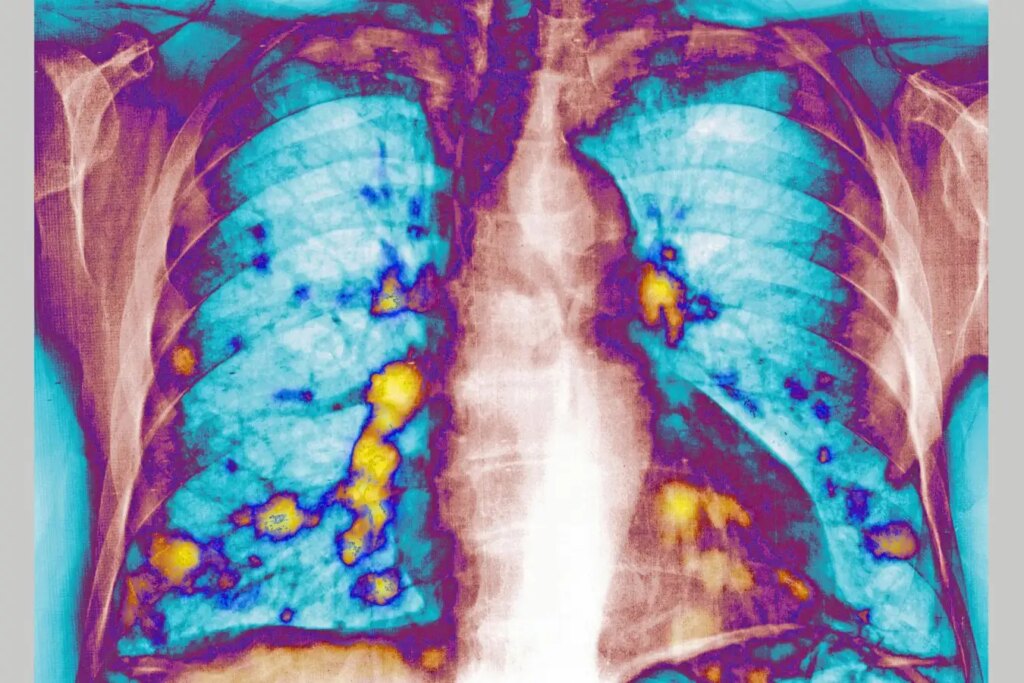
The lungs as a common site for cancer metastasis
Cavallini James/BSIP/Universal Images Group/Getty Images
Studies indicate that respiratory infections may temporarily inhibit the spread of cancer to the lungs
from other body parts. Recent experiments involving the respiratory syncytial virus (RSV)—known for causing cold-like symptoms and infecting nearly all children by age two—demonstrated its ability to hinder breast cancer cell colonization in the lungs. This suggests that the activation of infection-fighting proteins during such infections might be leveraged for therapeutic purposes.
According to research, the majority of cancer fatalities result from the metastasis of tumor cells from their original locations. Although early-stage cancer can often be treated effectively through surgery or radiation, once it metastasizes, treatment challenges increase significantly, with low success rates. As highlighted by David Withers from the University of Oxford, this presents a major clinical hurdle.
The implications of respiratory infections, including influenza and COVID-19, on cancer metastasis are captivating researchers, especially given the lungs’ susceptibility as a common site for such spread.
In mouse studies, RSV successfully triggered an immune response that inhibited the migration of breast cancer cells to the lungs. Cecilia Johansson from Imperial College London remarked, “This finding is remarkable, as it represents a novel aspect of viral impact on cancer.”
The research team infected 23 mice with RSV, while 16 healthy mice were used as controls. Following a 24-hour period, all mice received injections of breast cancer cells. After 28 days, lung tumor nodules in the RSV-infected mice were reduced by 65 to 70 percent compared to controls.
However, the size of the nodules that formed remained similar in both groups, indicating that the virus did not significantly impede cancer cell proliferation once they were established in the lungs. This aligns with previous studies showing that the viruses associated with swine flu and COVID-19 could activate dormant cancer cells after they have metastasized to the lungs.
Johansson and her team subsequently explored the role of type I interferon, a protein within lung cells that inhibits viral replication. Their findings revealed that this protein makes it considerably more challenging for cancer cells to develop new tumors.
To determine whether administering type I interferon could replicate the effects of RSV, researchers provided another group of mice with interventions, receiving doses of interferon prior to cancer cell injection. Initial results suggested that interferon was “slightly” more effective than the viral infection at limiting tumor cell ingress into the lungs.
“The study indicates that the type I interferon response, typical of acute viral infections, induces significant alterations in the lung epithelium,” explained Withers. “Although still in the preclinical phase, these results uncover exciting avenues to potentially enhance patient protection against metastasis.”
Researchers speculate that multiple mechanisms contribute to how type I interferon prevents tumor cell dissemination. Their focus remained on one particularly impactful method involving galectin-9, a protein produced in response to these interferons.
Johansson is optimistic that these findings could eventually lead to new drugs designed to thwart the spread of breast cancer and other tumors to the lungs. She stated, “Despite being early preclinical findings, we can test these strategies to develop new treatments against cancer without relying on viral infections.”
Claire Bennett from University College London did not participate in the study.
While the theoretical approach posits that the same strategy could thwart cancer spread to the lungs from various body sites, Johansson underlines the need for further investigation. She emphasizes that delivering type I interferon intranasally may create harmful airway inflammation.
The research team aims to conduct additional studies to deepen the understanding of interferons’ effects on cancer propagation. “We aspire to explore whether we can emulate this effect using IFN-inducing agents and how to effectively target the lung epithelium and stroma,” Johansson concluded. “Our goal is to eventually translate these findings into human studies and identify therapeutic targets in clinical settings, though that lies ahead.”
Topics:
Source: www.newscientist.com